Protomer Gets Boost to Develop Glucose Responsive Insulin
Francesca Billington is a freelance reporter. Prior to that, she was a general assignment reporter for dot.LA and has also reported for KCRW, the Santa Monica Daily Press and local publications in New Jersey. She graduated from Princeton in 2019 with a degree in anthropology.
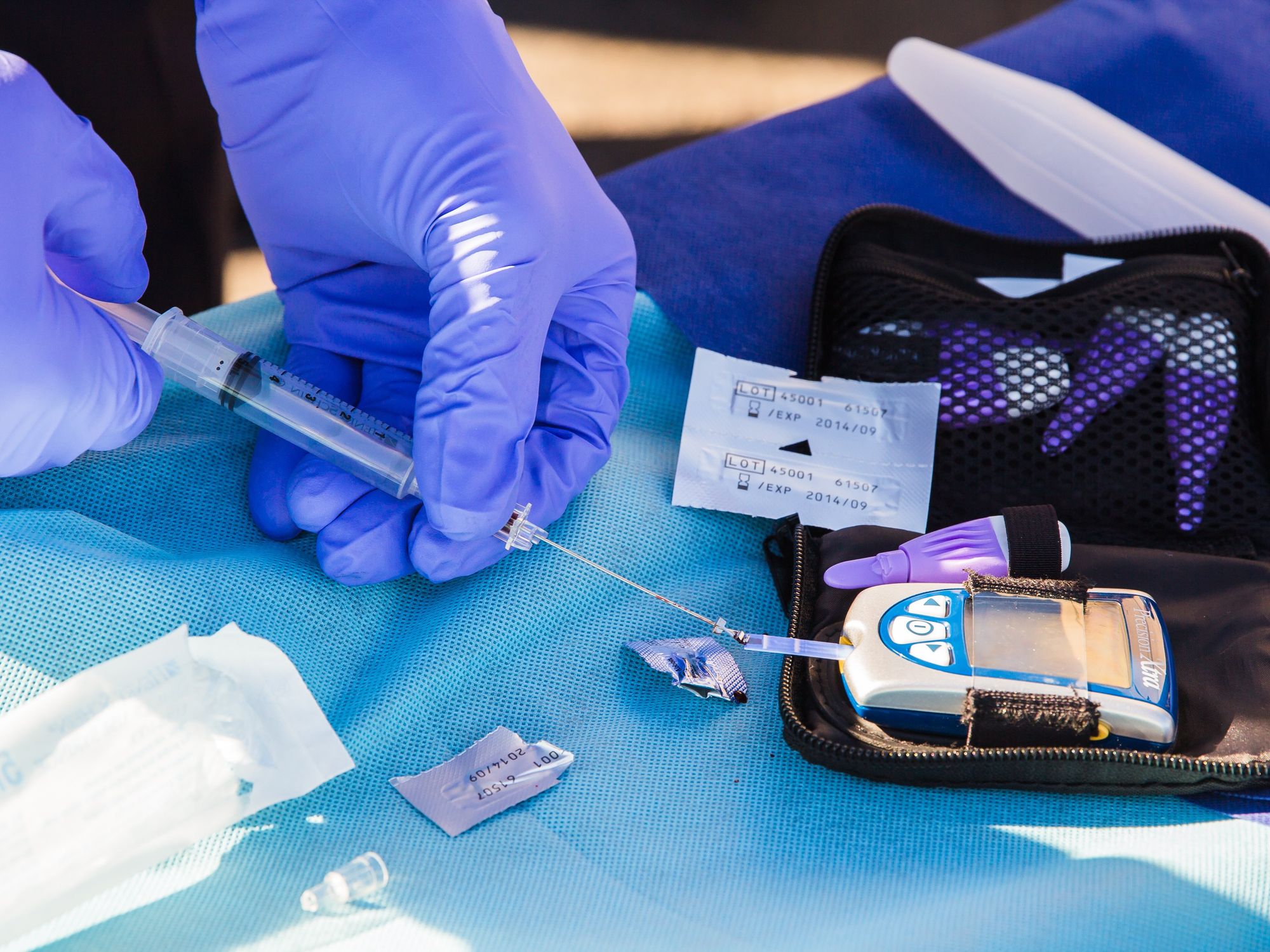
Protomer Technologies Inc., a Pasadena-based biopharmaceutical startup, closed a second round of funding to expand the tech platform it's using to develop insulin that adjusts according to a diabetes patient's blood sugar levels.
The undisclosed raise came from the JDRF T1D Fund, a Boston-based venture philanthropy fund that focuses on type one diabetes research.
The pre-clinical stage startup was launched in 2015 by a team of Caltech faculty and alumni to build a "heavy chemistry-based platform," said CEO and founder Alborz Mahdavi, who received his PhD in bioengineering from the university. They're now using the technology platform to create drugs that can activate once they've been injected into patients, including a new type of insulin.
The treatment still needs FDA approval.
Typically, diabetes patients carry with them glucose meters and insulin to track and maintain the sugar levels in their blood. Protomer's product is designed to activate itself automatically depending on a person's blood sugar levels.
"With this insulin, you don't need to worry about that," Mahdavi said. "There's enormous interest in this. This insulin will be completely transformative for people with diabetes."
Mahdavi said the platform could also be applied to other therapeutic settings like neuroscience and oncology. For example, a drug injected in patients with cancer could switch off if the treatment isn't working. It could also activate in a specific region of the body, which means that side effects of chemotherapy would diminish if the drug were "only active locally."
"Imagine you have pancreatic cancer and you're taking an oncology drug," he said. "The problem is that the drug will be active all over the body, which is why you get all these side effects."
Dave Whelan, CEO of BioscienceLA, said several companies in the field are engineering cells designed to fight cancer cells this way. It's a challenge researchers have been trying to crack for a while.
"It's a long time coming," he said. "It's very reassuring to see investments in this space because the last several months there's been so much focus on COVID, and part of that has been at the expense of other disease areas."
As the pandemic ushers in a new way of telemedicine, he sees an urgency in introducing therapeutics that don't require in-person physician attention. Meanwhile, cancer patients and certain diabetes patients may be more at risk if they contract the virus.
"When you see something like this, it just makes me feel good that there's still attention being paid to these diseases and conditions that will be with us long after COVID," he said.
Protomer's first equity investment, also an undisclosed amount, was led by pharmaceutical giant Eli Lilly and Company. Whelan said that move might signify a future acquisition, as big pharma often looks for innovation outside their own companies.
"It's not only an advance for life sciences and care, but because they're making progress and getting additional funding, that helps them grow the whole industry here," Whelan said. "We absolutely need more of that going on in L.A."
Clarification: An earlier version of this post stated that the T1D Fund has spent over $2 billion on type one diabetes research. While its nonprofit parent company JDRF may have spent that much, the T1D Fund has not.
Francesca Billington is a freelance reporter. Prior to that, she was a general assignment reporter for dot.LA and has also reported for KCRW, the Santa Monica Daily Press and local publications in New Jersey. She graduated from Princeton in 2019 with a degree in anthropology.